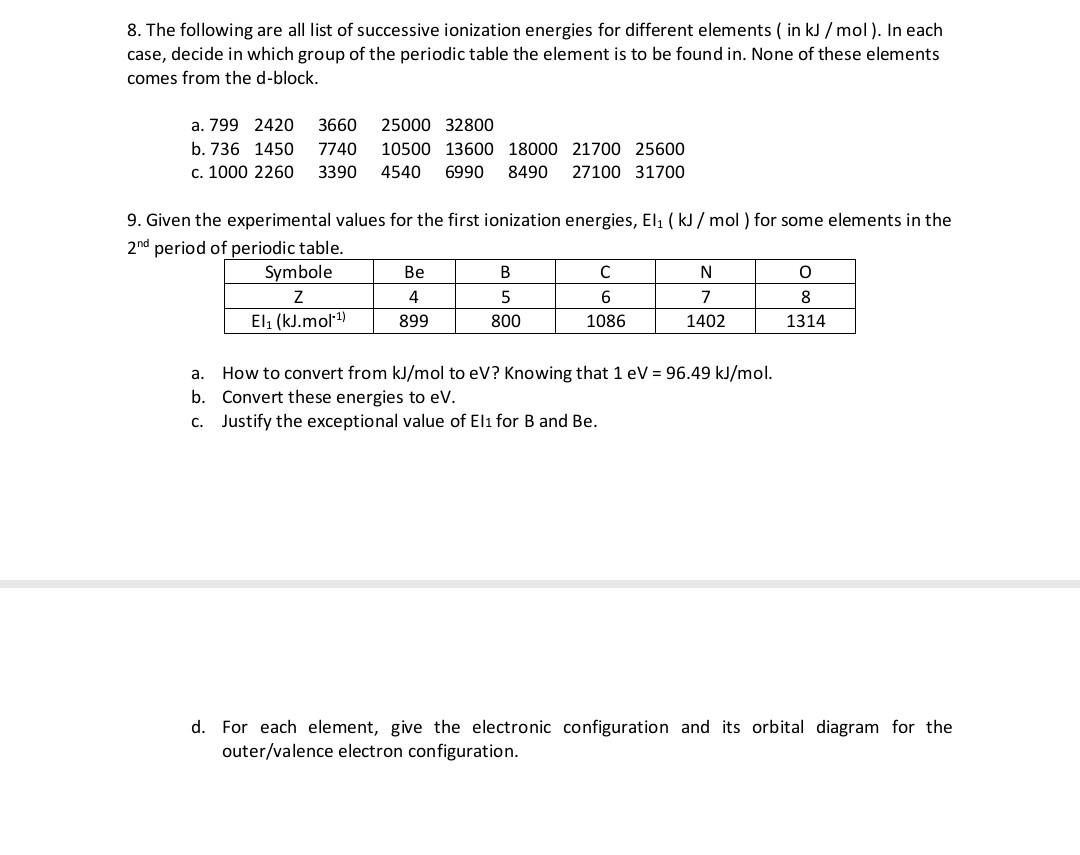
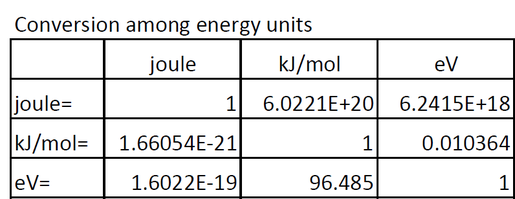
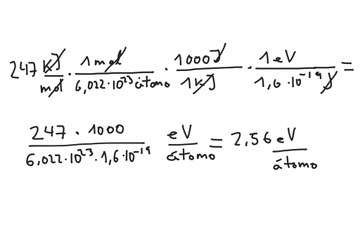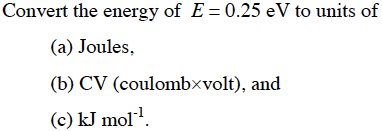Week 1: Basics Reading: Jensen 1.6,1.8,1.9. Two things we focus on DFT = quick way to do QM – 200 atoms, gases and solids, hard matter, treats electrons. - ppt download

SOLVED: Calculate (a) the energy (in eV) of a 5.3-Ã… X-ray photon and (b) the energy (kJ/mol) of a 530-nm photon of visible radiation. Given the following constants and unit conversion factors:

1 MODELING MATTER AT NANOSCALES 6. The theory of molecular orbitals for the description of nanosystems (part II) The Hartree-Fock method applied. - ppt download

SOLVED: (a)how much energy in KJ/mol is released when an electron makes a transition from n=5 to n=2 in a hydrogen atom?

Thermodymanics Lecture 3 8/31/2004. Units Energy Ultimate source of energy is the sun E = h 57 Kcal/mol of photons green light or 238 KJ/mol 1 cal = - ppt download

Table 2 from A mathematical introduction to Hartree-Fock SCF methods in Quantum Chemistry | Semantic Scholar