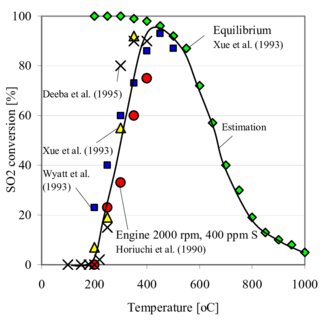
SO2 - SO3 conversion for set 1. Explanation: Δ… SO3 concentration in... | Download Scientific Diagram

Unprecedented Ambient Sulfur Trioxide (SO3) Detection: Possible Formation Mechanism and Atmospheric Implications | Environmental Science & Technology Letters

The value of heat of formation of SO2 and SO3 are −398.2 kJ and −198.2 kJ. The heat of formation of this reaction will be SO2+12O2→SO3
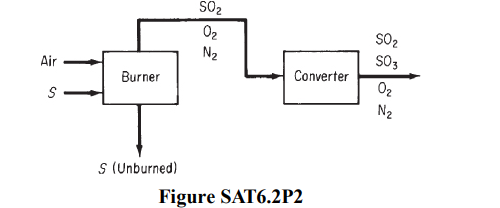
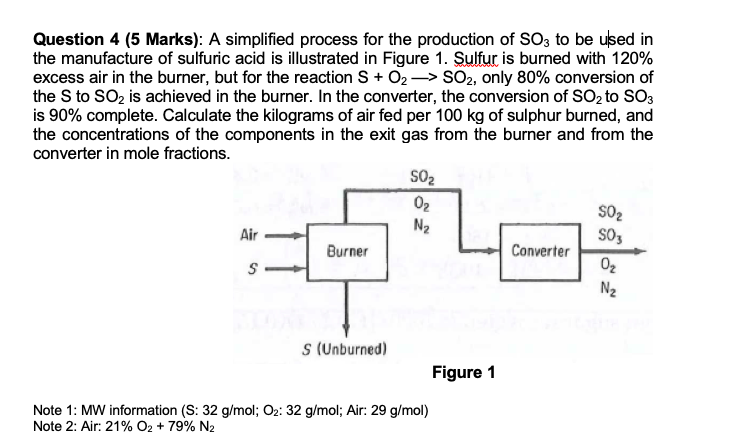
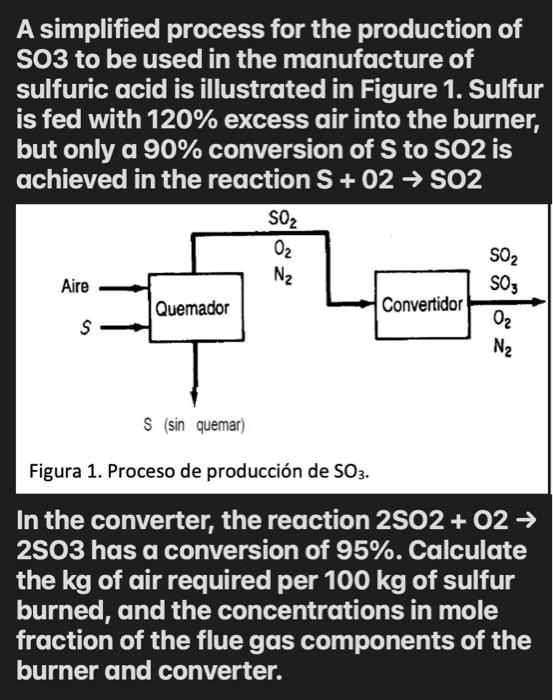
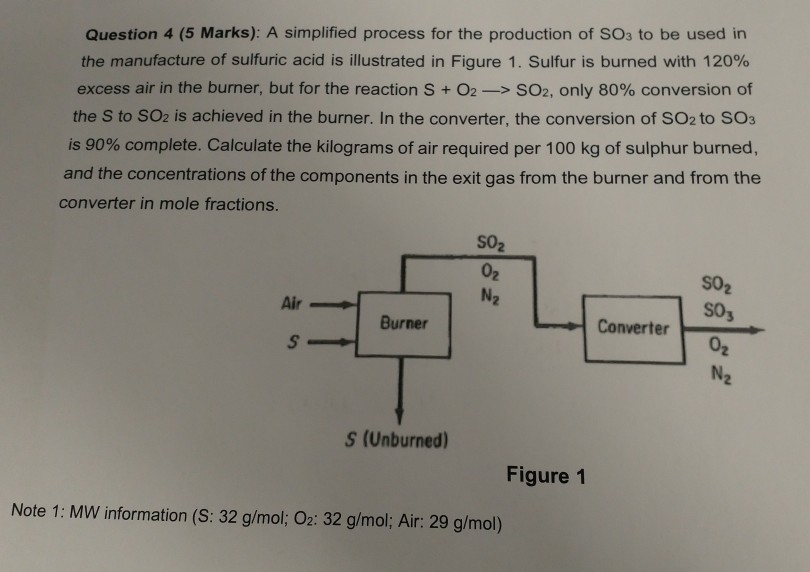
Solved) - A simplified process for the production of SO3 to be used in the... (1 Answer) | Transtutors

Reverse Conversion Treatment of Gaseous Sulfur Trioxide Using Metastable Sulfides from Sulfur-Rich Flue Gas | Environmental Science & Technology

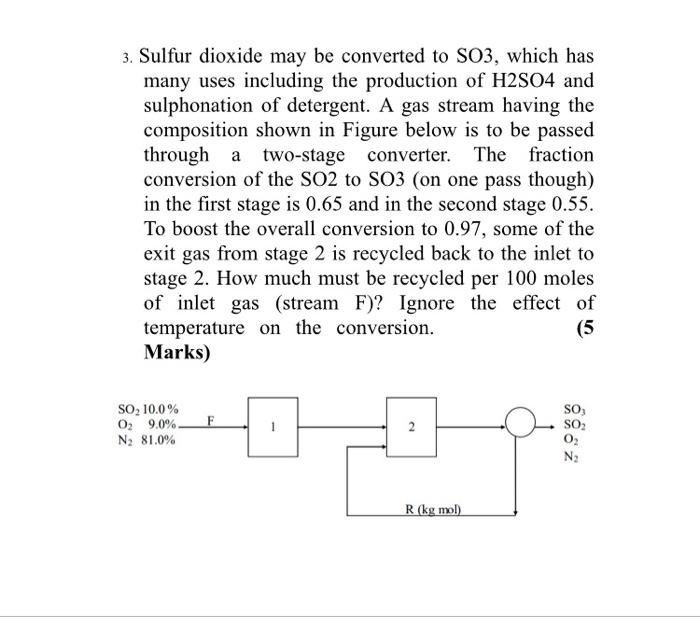
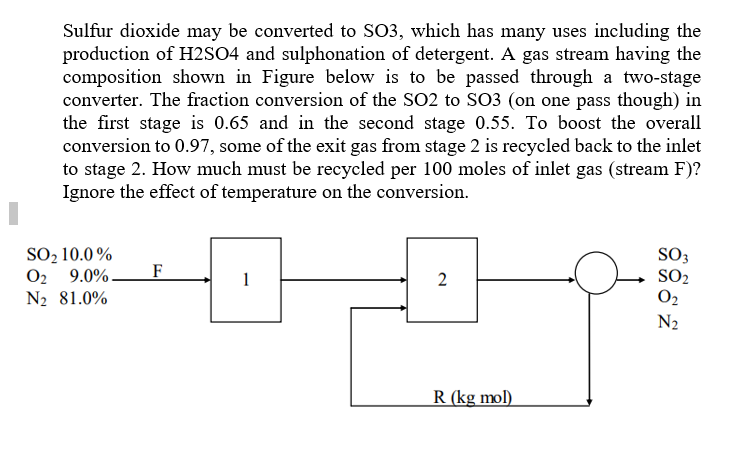
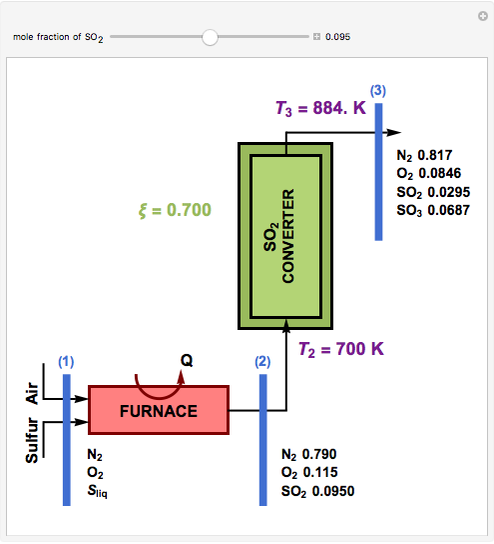
SOLVED: Sulfur dioxide may be converted to SO3, which has many uses including the production of H2SO4 and sulfonation of detergent. A gas stream having the composition shown in Figure below is

![Assamese] Name the catalyst used for conversion of SO2 to SO3 in the Assamese] Name the catalyst used for conversion of SO2 to SO3 in the](https://static.doubtnut.com/ss/web-overlay-thumb/8113909.webp)